EU-CTR
Automate your EU-CTR compliance workflows for faster, error-free clinical submissions.
AI Skills: Conversation
Princeton Blue’s EU-CTR solution aims to streamline and automate the internal workflow process to create a set of documents for submission/transition under the new EU-CTR guidelines.
Watch Solution Demo
Please tell us about yourself

The new version, powered by Appian AI is now available.
Business Drivers for Regulatory Submissions
In January 2022, the European Union introduced a new Clinical Trials Regulation (CTR), repealing the earlier Directive 2001/20/EC, or CT Directive (CTD). The new EU-CTR regulation aims to harmonize the process of assessment and supervision of clinical trials throughout the EU.
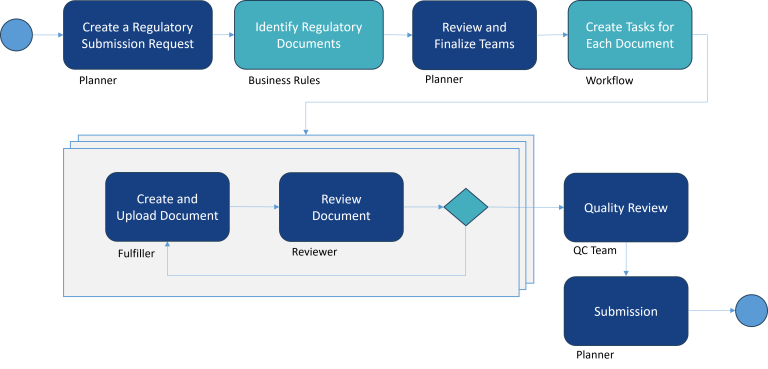
All Pharma companies operating in the EU must comply with EU-CTR for all new trials. Each submission requires careful identification of country-specific documents, and a workflow cycle that spans creation thru approval.
These submissions are typically a set of documents created by various teams within the regulatory group of the pharmaceutical company.
- These documents need to be created by personnel from various teams, reviewed, approved, and submitted in a timely manner.
- Each document must be thoroughly reviewed and approved before submission.
- Compliance requirements must be met with detailed audit trial and documentation.
Watch Solution Demo
Please tell us about yourself

The new version, powered by Appian AI is now available.
Challenges with the Submissions Process
Limitations of rudimentary tools: Often, the regulatory submission process is executed and tracked using rudimentary tools like Excel, SharePoint, and Email, limiting the flexibility in operations, and leading to lack of efficiency.
No process transparency: Since a submission request can span multiple countries, rudimentary tools do not provide the visibility into the latest submission status, thereby limiting the regulatory team’s ability to take effective decisions, potentially delaying the regulatory submission.
Streamlining the Internal Workflow for EU-CTR Submissions
Princeton Blue’s EU-CTR solution automates the processing of creating and approving various types of regulatory documents using the Appian Low-code Process Automation technology.
- The solution orchestrates the entire workflow from document identification to approvals until it is ready to be submitted to the CTIS.
- Automates the identification of Part 1 and Part 2 documents.
- Integrates and extends your existing applications that support regulatory operations to bring data and documents into the context of the process.
- It provides an agile and modern user experience across different devices and form factors.
- Provides insight into the submissions process via interactive graphical dashboards and reports and helps quickly identify and resolve bottlenecks.
- The solution is also easily customized and adapts to your needs.
Key Highlights of the Solution

Intelligent Document Processing

Configurable Business Rules
Validate incoming orders, identify potential exceptions, and proactively address issues such as projected inventory stockouts, warehouse capacity bottlenecks, and lead time shortages.
Flexible Workflows

Regulatory Compliance

Mobile-Enabled Modern UX

Interactive Dashboards and Reports
Key Highlights of the Solution

Intelligent Document Processing

Configurable Business Rules

Flexible Workflows

Regulatory Compliance

Mobile-Enabled Modern UX

Interactive Dashboards and Reports
Key Features of the Solution
- Automation of country-specific document identification via configurable business rules
- Customizable workflows for defining and approving a submission request followed by subsequent life cycle of each document creation, approval, and finalization.
- Integration with existing systems to pull and push data needed for the regulatory submission.
- Automation of document organization with a Document Repository for easy retrieval and offline search.
- Identification of potential bottlenecks and helps manage SLA compliance with active process monitoring.
- Detailed audit-trail for compliance with regulatory requirements and driving accountability.
- Superior user experience across all devices, form factors and platforms with a mobile-enabled modern design
- In-depth insight into the regulatory submission operations via interactive graphical dashboards and reports.
- Flexibility to easily make changes to execution parameters such as priorities, owners, due dates, and more, driven by the needs of the business.

Enhanced Efficiency

Improved Trial Timelines
By effectively managing the supply chain and addressing challenges such as unpredictable lead times, stockouts, and warehouse capacity bottlenecks, the solution helps maintain the planned timelines of clinical trials. Avoiding delays ensures the timely completion of trials, accelerating time to market for new drugs.

Greater Compliance
The solution incorporates configurable business rules and maintains a detailed audit trail, enabling pharmaceutical companies to comply with complex GxP and regulatory guidelines. Mitigate non-compliance risks and associated costs, ensuring adherence to local regulatory requirements throughout the clinical supplies shipping process.

Enhanced Visibility

Reduced Costs

Superior User Experience
Key Features of the Solution
Process Improvement
- Automation of Part 1 and Part 2 document identification via configurable business rules
- Customizable workflows for defining and approving documents.
- Flexibility to easily make changes to execution parameters such as priorities, owners, due dates, and more, driven by the ever-changing business needs.
Process Monitoring and Compliance
- In-depth insight into the submissions process via interactive graphical dashboards and reports.
- Proactive identification of potential bottlenecks to ensure SLA compliance with active process monitoring.
- Detailed audit-trail for compliance audits and driving accountability.
Integration
- Integration with existing systems to pull and push data as and when needed in the process.
- Automation of document organization with a Document Repository for easy retrieval and offline search.
User Experience
- Intuitive user experience across all devices, form factors and platforms with a mobile-enabled modern design.
- Easily customized and adapted to your needs.
AI-Powered
- Appian AI to analyze structured data of Submissions
- Appian AI Copilot to answer questions about EU-CTR Submissions and related record data, enabling managers to interact with conversational AI and get quick insights