The Drug Safety solution augments existing systems such as Argus, Veeva and Medidata to unify data and automate the workflow experience for effective analysis and prioritization.
Powered by Low-code technology, it helps pharma companies focus on patient safety with a modern, mobile-enabled app for seamless collaboration, multi-channel notifications, real-time dashboards, and SLA monitoring.
Watch Solution Demo
Please tell us about yourself
Key Drivers for Drug Safety
After going to market, drug companies are required to ensure that their product continues to be safe and effective. They also need to demonstrate that a rigorous and ongoing safety surveillance program is in place to support quick action on any safety concerns. Here are the key drivers to ensure drug safety success.

Timeliness of Response
Each case must be responded to in a timely manner

Configurable Workflow
Flexibility in defining steps dynamically for each case

Collaboration
Active collaboration between all stakeholders

Progress Visibility
Real-time visibility into status of each case

SLA Compliance
Compliance to statutory timelines is critical

Drug Safety Process Pain Points
The Drug Safety process is often burdened with rigid workflows, lack of transparency into the case, silos created by disparate systems, and lack of effective collaboration among the internal and external stakeholders.
While every organization strives to have processes in place for Drug Safety, most find that:
- Technology is not flexible enough to support each case differently
- Lack of agility to keep up with ever-changing business needs
- Limited transparency created by siloed systems
- Unorganized collaboration based on phone, email, and MS Office
- Lack of SLA monitoring and Audit Trail records
Streamlined and Collaborative Emergency Response
Powered by BPM and Low-code
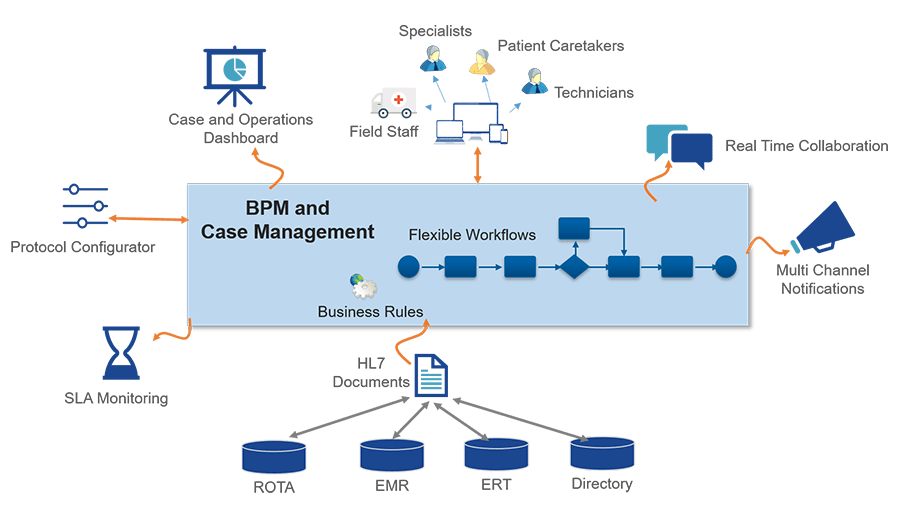
The Dynamic Emergency Response solution addresses the pain points by automating the process with the BPM and Low-code platform. The Protocol Configurator provides a flexible mechanism to dynamically configure the sequence of tests and consultations needed to address each type of emergency.
It provides the ER team with a modern, mobile-enabled app for seamless collaboration, multi-channel notifications, HL7 integration with EMR systems, real-time dashboards and SLA monitoring.
Streamlining the Drug Safety Process
The Drug Safety solution augments existing systems such as Argus, Veeva and Medidata to unify data and automate the workflow experience for effective analysis and prioritization.
Using dynamic workflows, real-time collaboration, and a full transparent view into the process, your team can respond to adverse events swiftly while reporting to the health authority in a timely and accurate manner.
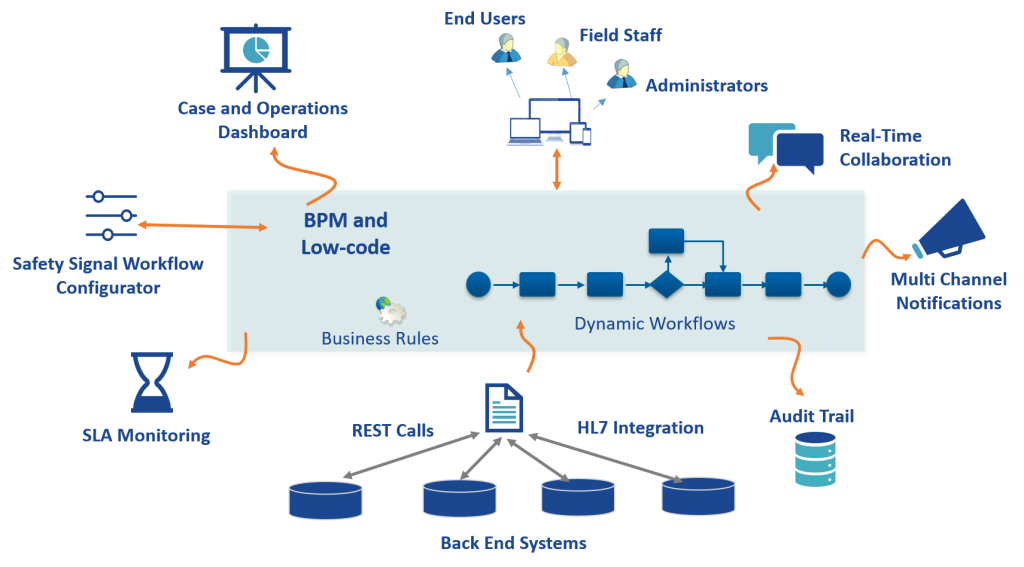
The Drug Safety solution addresses the pain points by automating the process with a Low-code platform. The Safety Signal Workflow Configurator provides a flexible mechanism to dynamically configure the most effective activities for signal analysis and prioritization. It provides the organization with a modern, mobile-enabled app for seamless collaboration, multi-channel notifications, real-time dashboards, and SLA monitoring.
A safety signal workflow is a pre-configured set of activities that need to be performed for a specific type of drug safety issue. The Drug Safety solution allows each workflow to be completely tailored for each drug safety report based on the unique condition.
The Drug Safety solution provides configurable and dynamic workflow, improves timeliness and compliance, and allows for real-time collaboration among the stakeholders with the objective of improved transparency in processing the safety signal, and timely and accurate reporting to the health agency.
Key Highlights
The Dynamic Emergency Response solution allows an Emergency case to be handled in a configurable and dynamic manner based on hospital protocols, staff availability and individual needs of the patient. This improves compliance, timeliness and allows for real-time collaboration between the patient’s care givers, EMT, doctors and hospital staff.
Timeliness
Every minute can save a life
Configurable Protocols
Dynamic workflows based on type of emergency
Collaboration
Active collaboration between all stakeholders
Progress Visibility
Real-time visibility into status of patient care journey
Statutory SLA Compliance
Accurate monitoring and reporting of SLA compliance
Real-Time Dashboard
High-level overview of emergency cases and their status, through various interactive graphs
Key Highlights
The Dynamic Emergency Response solution allows an Emergency case to be handled in a configurable and dynamic manner based on hospital protocols, staff availability and individual needs of the patient. This improves compliance, timeliness and allows for real-time collaboration between the patient’s care givers, EMT, doctors and hospital staff.

Timeliness
Every minute can save a life

Configurable Protocols
Dynamic workflows based on type of emergency

Collaboration
Active collaboration between all stakeholders

Progress Visibility
Real-time visibility into status of patient care journey

Statutory SLA Compliance
Accurate monitoring and reporting of SLA compliance

Real-Time Dashboard
High-level overview of emergency cases and their status, through various interactive graphs
Key Highlights
The Dynamic Emergency Response solution allows an Emergency case to be handled in a configurable and dynamic manner based on hospital protocols, staff availability and individual needs of the patient. This improves compliance, timeliness and allows for real-time collaboration between the patient’s care givers, EMT, doctors and hospital staff.

Configurable Protocols
Dynamic workflows based on type of emergency

Timeliness
Every minute can save a life

Collaboration
Active collaboration between all stakeholders

Progress Visibility
Real-time visibility into status of patient care journey

Statutory SLA Compliance
Accurate monitoring and reporting of SLA compliance

Real-Time Dashboard
High-level overview of emergency cases and their status, through various interactive graphs
Key Highlights

Configurable Workflows

Signal Visibility

Collaboration

Interactive Dashboards

Flexible Business rules
Watch the Drug Safety Solution in Action
Key Highlights

Configurable Protocols

Timeliness

Collaboration

Progress Visibility

Statutory SLA Compliance

Real-Time Dashboard