As the pharmaceutical industry increasingly finds itself adapting to changes in competitive and regulatory pressures, the need for an efficient and accurate Regulatory Information Management (RIM) solution continues to rise. Combining the ability to consolidate regulatory metadata from across the organization with features to manage, track, and report – a good RIM solution brings business efficiency and data confidence to an organization.
Need for Regulatory Information Management (RIM) Solution
Historically, pharmaceutical companies have attempted to tackle this challenge by building custom database solutions that are expensive and hard to maintain. Alternatively, they struggle with a collection of cumbersome and unreliable Excel workbooks that may buckle under health authority scrutiny. The time for a sustainable and straightforward technology option is paramount.
Princeton Blue, in partnership with Appian’s award-winning custom application software, is leading the charge to deliver a full set of RIM components that can be easily individualized to provide a tailored solution. The key strength of the RIM is a flexible framework architecture that delivers reliability, scalability, and low-cost maintenance.
This solution will be rolled out over the course of the next year as a collection of modules targeted to address different aspects of the RIM portfolio. This approach allows for modules that can be used independently to target issue areas, or linked together to create a full umbrella RIM solution.
The first module that we will focus on is Data Governance. As pharmaceutical companies continue to grow the size of their data warehouses management of data, as well as integration across applications has become vital.
The model for a RIM strategy road-map developed by Gen and Associates, starts with Master Data Governance and ends with a great User Experience. The middle layer (solution sets) is what most companies already have in place, but Princeton Blue, partnered with Appian, can help you build a better complete solution ready for the challenges of tomorrow.
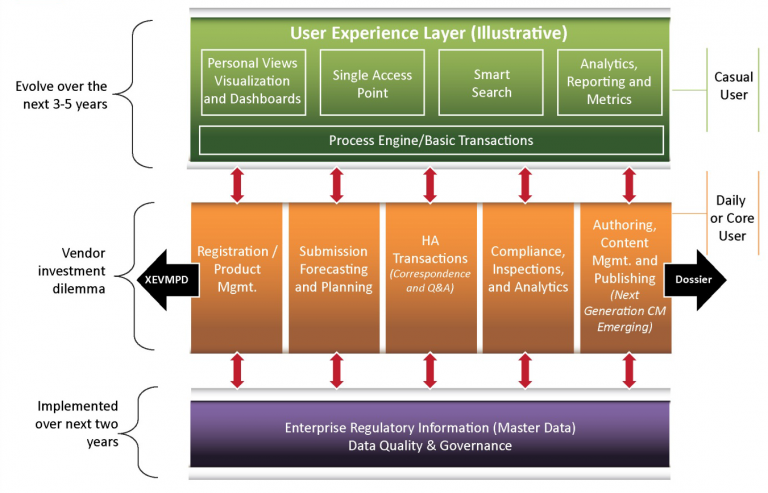
Princeton Blue’s World-Class RIM Solution
The process of managing Master Data changes requires:
- Diligent tracking of the “conversation” that occurs throughout the change request and approval process
- Easy-to-learn user interface for changing/creating new business rules around required approvals
- Sophisticated user experience for viewing complete data related to Master Data change requests
Here is how Princeton Blue’s World-Class RIM Solution can help you accomplish all of the aspects mentioned above by leveraging the power of Appian:
-
The audit trail throughout the change request is maintained in multiple ways:
- The Appian News Feed allows users to easily post comments in reference to any event
- A Notes section in Records facilitates a conversation that is persisted to your database and can even be accessed outside of Appian
- An approval log is kept in the database as wet signatures are obtained throughout the approval process
-
New business rules are easy to update and maintain
- By leveraging the power and flexibility of the Princeton Blue’s Advanced Rules Management solution, the ability to change business rules is taken away from the developers and given back to the business users
-
Any application is only as good as its user experience, and you will have satisfied end-users when the application is built using the Appian framework
- Records allow users to easily see all Master Data change requests that have gone through the system and allow for drilling down to see the details of each request
- Robust Reports for end-users to see a summarized view of all requests by making use of visuals and easy-to-use filters to manipulate data into a usable format
- Easy-to-understand Appian SAIL forms are used throughout the process as tasks move along from one group in the organization to the next
Let’s take the next step and start building a complete RIM solution that can help your organization tackle the regulatory challenges of the future. What better way to start than with the base of a good RIM strategy road-map: Data Governance. Please visit our website at www.princetonblue.com and request to have us contact you, or you may reach out to us directly at (908)369-0961.
Feel free to share your thoughts in the comment section below. Follow Princeton Blue on Facebook, LinkedIn, Twitter and Google+ to get key BPM-specific insights on a regular basis.



